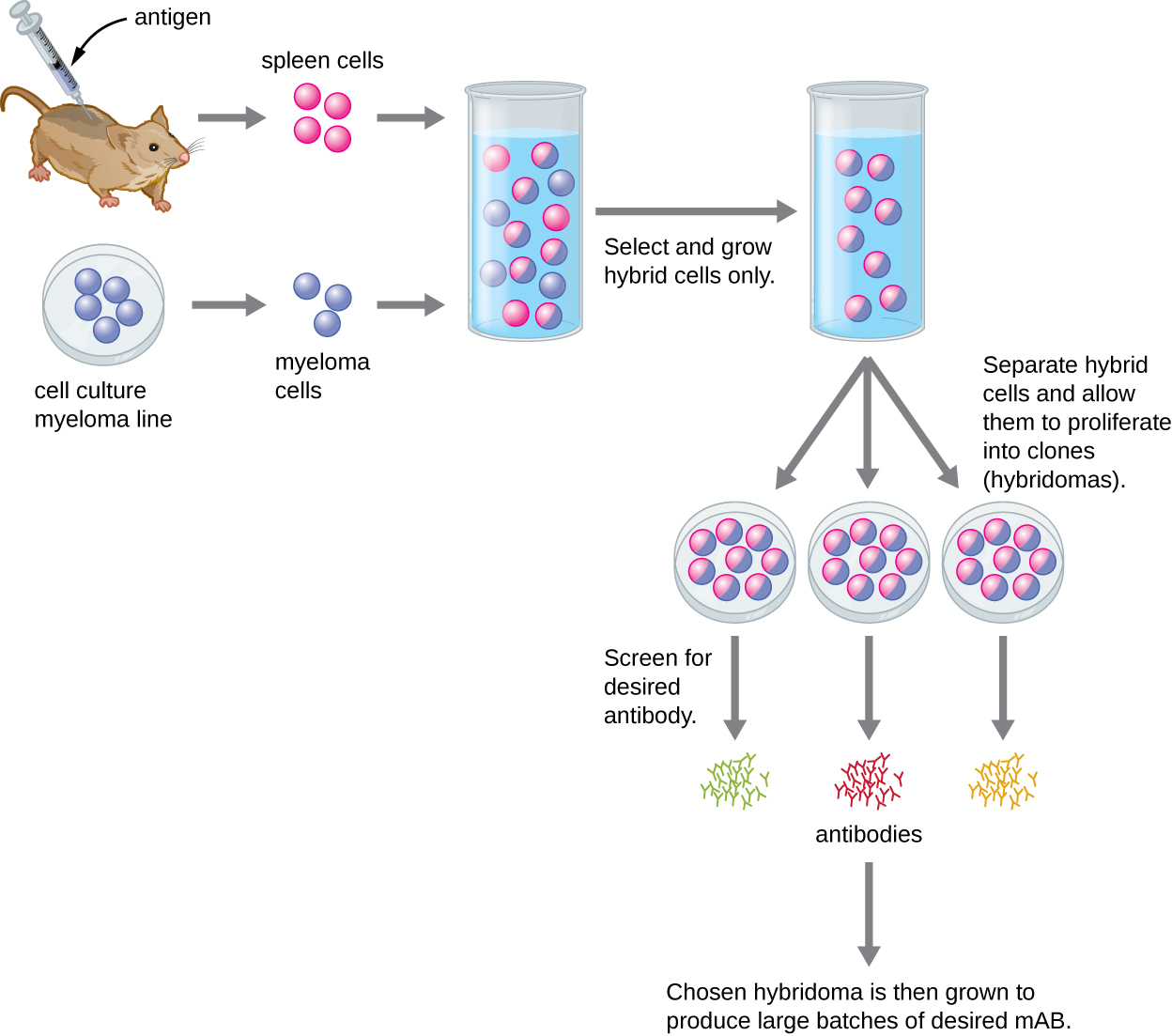
 pastoris is also a commonly used microbial system that can be engineered to produce both full-length human-like glycosylated antibodies and antibody fragments. Presently, this system is also well-established and the preferred method for the production of camelid’s heavy chain antibody fragments. cerevisiae was the first microbial system to be employed in the production of recombinant biopharmaceuticals. There are several advantages in using antibody fragments, namely, the decreased production cost (inexpensive culture media) and lead time, increased ease of genetic manipulation in comparison to mammalian systems and increased tissue penetration. single-chain antibody fragments – scFvs). Thus, microbial systems are increasingly gaining traction for the production of antibody fragments (e.g. However, functional Fab and Fv regions suffice to bind and recognize antigens. For these reasons, researchers are increasingly interested in alternative antibody production methods.Īntibody production steps in microbial systemsīacteria, such as Escherichia coli, and yeast systems, such as Saccharomyces cerevisiae and Pichia pastoris, are unable to perform human-like glycosylation. Moreover, mammalian systems are hard to scale-up, requiring long and incremental scale-up steps which generate increased processing times. These cells require the use of expensive media and they are highly susceptible to viral and bacterial infections which may hamper long-term manufacturing processes. While transient systems such as embryonic kidney (HEK 293), amniotic (CAP), a hybrid of HEK 293 and lymphoma (HKB-11), and embryonic retina (PER.C6) are preferably used for preliminary studies.ĭespite the technological advances of the last decade, the use of mammalian systems, especially stable systems, still has a very high production cost. For this reason, the generation and subsequent selection of stable CHO germlines is usually time-consuming, technically-demanding and, for this reason, it is reserved for large-scale commercial antibody production. However, the culture medium and conditions of these cell lines need to be carefully optimized to maximize and stabilize antibody production. Many antibodies approved for therapy have been obtained from recombinant production in CHO cells. The use of these cell lines presents several advantages including its relatively simple and human-like glycosylation process, fast growth rates and high antibody production titers, ease of genetic manipulation, good proliferation in large-scale suspension cultures and adaptability to protein-free medium. Traditionally, Chinese hamster ovary (CHO) cell lines have been the preferred stable system for recombinant antibody production. That is why, historically, the use of mammalian cell lines able of human-like glycosylation has played a dominant role in antibody production. Most non-human expression systems either cannot perform post-translational glycosylation or they present very distinct glycosylation patterns when compared to human cell lines. 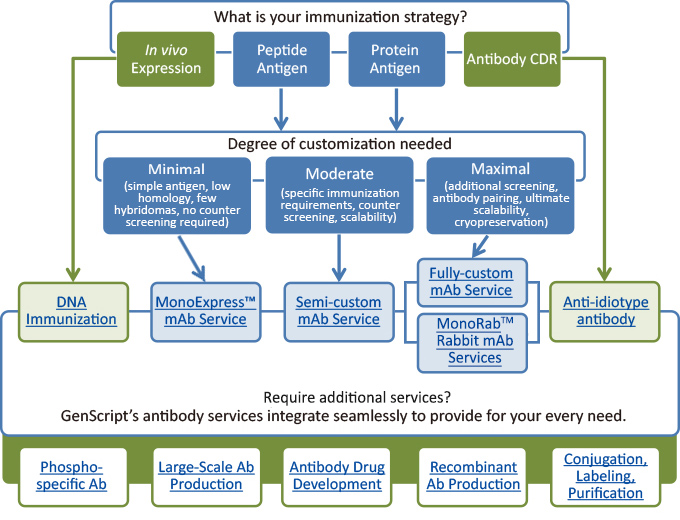
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
